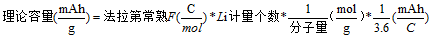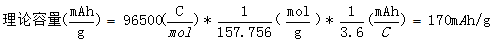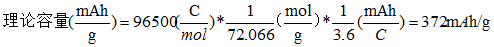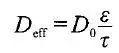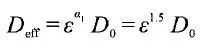|
|
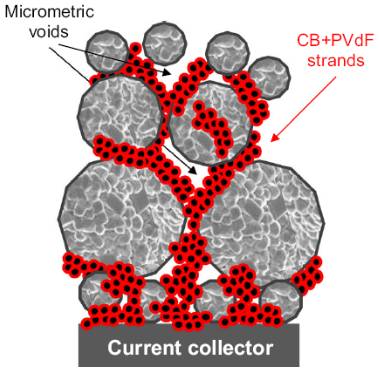
Lithium-ion battery is a high-capacity and long-life environmentally friendly battery with many advantages, which is widely used in energy storage, electric vehicles, portable electronic products and other fields. The electrode electrode is the basis of lithium-ion power batteries, which directly determines the electrochemical performance and safety of the battery. Lithium battery electrodes are a coating composed of particles that is uniformly applied to the metal current collector. The lithium-ion battery electrode coating can be seen as a composite material, as shown in Figure 1, which consists of three main parts: (1) Active substance particles; (2) The component phase (carbon gel phase) where the conductive agent and the binder are mixed with each other; (3) Pore, fill the electrolyte. The volume relationship of each phase is expressed as:Porosity + Volume Fraction of Living Matter + Volume Fraction of Carbon Gel = 1 (1) First, lithium battery pole piece design The design of lithium battery electrodes is very important, and the basics of lithium battery electrode design are briefly introduced. (1) The theoretical capacity of the electrode material The theoretical capacity of the electrode material, that is, the capacity that can be provided by assuming that all the lithium ions in the material participate in the electrochemical reaction, the value of which is calculated by the following formula:
where Faraday's constant (F) represents the charge carried per mole of electrons in units C/mol, which is the product of the Avogadro number NA=6.02214 ×1023mol-1 and the metacharge e=1.602176 × 10-19 C, with a value of 96485.3383±0.0083 C/mol. For example, the molar mass of LiFePO4 is 157.756 g/mol, and its theoretical capacity is:
The ternary material NCM(1/1/1)(LiNi1/3Co1/3Mn1/3O2) has a molar mass of 96.461 g/mol and a theoretical capacity of 278 mAh/g The molar mass of LiCoO2 is 97.8698 g/mol, and if all the lithium ions are removed, its theoretical gram capacity is 274 mAh/g In the graphite anode, when the amount of lithium intercalation is the largest, a lithium-carbon interlayer compound is formed, with the chemical formula LiC6, that is, 6 carbon atoms combined with one Li. The mass of 6 C molar is 72.066 g/mol, and the maximum theoretical capacity of graphite is:
For silicon anodes, it can be seen from 5Si+22Li++22e- ↔ Li22Si5 that the molar mass of 5 silicon is 140.430 g/mol, and 5 silicon atoms are combined with 22 Li, the theoretical capacity of the silicon anode is: Material actual gram capacity = lithium ion intercalation coefficient × Theoretical capacity (3) (2) Battery design capacity and pole sheet density The battery design capacity can be calculated by equation (4):Among them, the areal density of the coating is a key design parameter, when the compaction density is unchanged, the increase of the surface density of the coating means that the thickness of the electrode piece increases, the electron transmission distance increases, and the electronic resistance increases, but the degree of increase is limited. In thick pole pieces, the increase in the migration impedance of lithium ions in the electrolyte is the main reason for affecting the magnification characteristics, considering the porosity and the twists and turns of porosity, the migration distance of ions in the pores is many times more than the thickness of the pole piece. (3) Negative-positive capacity ratio N/P The ratio of negative capacity to positive capacity is defined as:N/P = negative active material gram capacity×negative areal density×negative active substance content ratio÷ (positive active substance gram capacity× positive areal density × positive active substance content ratio) (5) N/P should be greater than 1.0, generally 1.04~1.20, which is mainly in a safe design, to prevent lithium ions on the negative electrode side without receiving a source and precipitation, design to consider process capabilities, such as coating deviation. However, when the N/P is too large, the irreversible capacity loss of the battery leads to low battery capacity and reduced battery energy density. For the lithium titanate anode, the positive electrode overdose design is adopted, and the battery capacity is determined by the capacity of the lithium titanate anode. The positive electrode overdose design is conducive to improving the high-temperature performance of the battery: the high-temperature gas is mainly derived from the negative electrode, and when the positive electrode is overdesigned, the negative electrode potential is lower, and it is easier to form an SEI film on the surface of lithium titanate. (4) The compaction density and porosity of the coating During the production process, the coating compaction density of the battery electrode piece is calculated by equation (6), Second, the basis of lithium battery raw material particles Lithium battery electrode is a powder particle composed of a coating, because the powder particle surface rough, irregular shape, in the stacking, particles and particles between there must be pores, and some particles themselves have cracks and pores, so the volume of the powder includes the volume of the powder itself, the pores between the powder particles and the pores inside the particles, therefore, the corresponding has a variety of electrode coating density and porosity representation. The density of powder particles refers to the mass per unit volume of powder. According to the volume of the powder, it is divided into three types: true density, particle density and bulk density. The various densities are defined as follows:a. True density refers to the density of the powder mass divided by the volume excluding the internal and external voids of the particles (true volume). That is, after excluding the volume occupied by all voids, the density of the substance itself is obtained. b. Particle density refers to the density of the powder mass divided by the volume of particles including open and closed pores. That is, the void between the particles is excluded, but the fine pores inside the particles themselves are not excluded, and the density of the particles themselves is obtained.c. Bulk density, that is, coating density, refers to the density of the powder divided by the volume of the coating composed of the powder. The volume used includes the pores of the particles themselves and the total volume between the particles.For the same powder, true density > particle density > bulk density.The porosity of the powder is the ratio of pores in the powder particle coating, that is, the ratio of the pores between the powder particles and the pores of the particles themselves to the total volume of the coating, which is often expressed as a percentage. The porosity of powder is a comprehensive property related to particle morphology, surface state, particle size and particle size distribution, and the size of its porosity directly affects the infiltration and lithium ion transport of the electrolyte. In general, the larger the porosity, the easier the electrolyte infiltration, and the faster the lithium ion transport. Therefore, in the design of lithium batteries, porosity is sometimes determined, and mercury pressure method and gas adsorption method are commonly used to measure. It can also be calculated by density calculation. When calculated with different densities, the porosity meaning is also different.When the density of living substances, conductive agents, and binders are calculated by true density, the calculated porosity includes the voids between particles and the internal voids of particles. When the density of living substances, conductive agents, and binders is calculated by particle density, the calculated porosity includes the voids between the particles, but not the internal voids of the particles. Therefore, the pore size of the lithium battery electrode piece is also multi-scale, generally the gap between the particles is in the micron level, and the internal void of the particles is in the nanometer to submicron level.In porous electrodes, the relationship between transport properties such as effective diffusivity and conductivity can be expressed by the following formula:
where D0 represents the intrinsic diffusion (conduction) rate of the material itself, ε is the volume fraction of the corresponding phase, and τ is the tortuosity of the corresponding phase. In the macroscopic homogeneity model, the Bruggeman relation is generally used, and the coefficient ɑ=1.5 is taken to estimate the effective physical properties of the porous electrode.
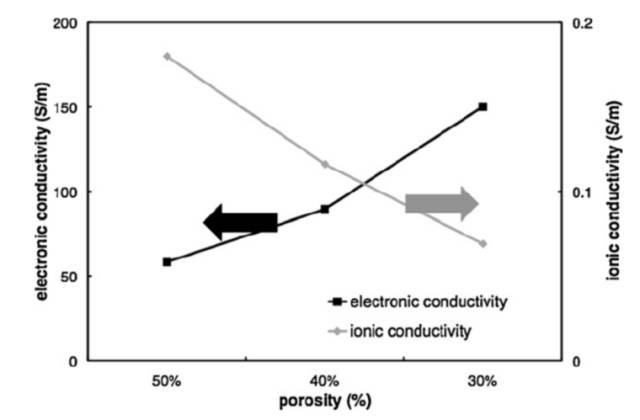
The electrolyte fills the pores of the porous electrode, and lithium ions are conducted through the electrolyte in the pores, and the conduction characteristics of lithium ions are closely related to porosity. The greater the porosity, the higher the volume fraction of the electrolyte phase and the greater the effective conductivity of the lithium ion. In the positive electrode piece, electrons are transported through the carbon gel phase, and the volume fraction of the carbon gel phase and the roundabout curvature of the carbon gel phase directly determine the effective conductivity of the electron. The volume fraction of porosity and carbon gel phase is contradictory, and large porosity inevitably leads to a decrease in the volume fraction of carbon gel phase, so the effective conduction characteristics of lithium ions and electrons are also contradictory, as shown in Figure 2. As porosity decreases, the effective conductivity of lithium ions decreases, while the effective conductivity of electrons increases. Balancing the two is also critical in electrode design.
Electron and lithium-ion effective conductivity vs. porosity Lastnews test 10Nextnews test 08 |