|
Under the same chemical system, the energy density can often be increased by optimizing the cell design parameters, such as: higher pressure solid density pole design, optimized conductive agent and electrolyte formulation. However, the increase in compaction density brings a series of problems, including the difficulty of electrolyte infiltration. If a certain design optimization is not carried out, the cell capacity and efficiency will be affected in the short term, and the cycle life and safety and reliability will be affected in the long run, so it is necessary to systematically study the key parameters that affect the speed of electrolyte infiltration. In addition, high-temperature cell wetting is also a key process affecting manufacturing costs, requiring extremely low dew point and high temperature environment, usually more than 24h. Therefore, considering the wetting rate from the design stage is also of great significance to reduce manufacturing costs. In this paper, the effects of different positive and negative compaction densities, different positive electrode current collectors, different positive electrode formulations, different infiltration temperatures and electrolyte formulations on the infiltration rate were systematically studied by building a suspension device to test the electrolyte infiltration climb rate. The use of the speed of electrolyte climb rate in the pole piece to judge the difficulty of electrolyte infiltration in cells of different designs has guiding significance for cell design. Experimentation The cathode selected for the experiment was the lithium iron phosphate cathode, the ratio of lithium iron phosphate, binder and conductive carbon black was 94.5%, 3.0% and 2.5%, the single-sided areal density was 164g/m2, and the design compaction density was 2.5g/cm3. The ratio of artificial graphite, binder, dispersant and conductive carbon black was 94.0%, 2.5%, 2.0% and 1.5%, the single-sided areal density was 76g/m2, and the design compaction density was 1.5g/cm3. The selected diaphragm for the experiment is a wet PE-based film. The solvent of the electrolyte is EC: DEC: DMC=2∶5∶3, the lithium salt is 1mol/L LiPF6, and the additive is 2% VC. The suspension method test electrolyte climb rate device is shown in Figure 1(a), and the operation process is as follows: place the device upright, inject electrolyte into it, the liquid level is slightly lower than the height of the green part of the shell, and cut the above sample to be tested into 20mm×260 mm strips. The bench dimensions are 55 mm (thickness)× 174 mm (length) × 200 mm (height), and the sample cutting length is recommended ≥ 1.2 times the shell height. The sample is fixed with double-sided tape in the grooves at the lower and lower ends of the separator shown in Figure 1(b), the fixing process needs to ensure that the sample is straightened without bending, and then the separator fixed with the sample strip is slowly placed into the housing, and the time and liquid level are recorded to correspond to the scale. The left side of each groove of the separator is designed with a scale of 0.5 mm, which serves as the starting point for the immersion experiment, and then slowly removes the pipette and seals the injection hole with a rubber stopper. Subsequently, the height ΔH of the climbing electrolyte in the pole piece is recorded at certain intervals, and the corresponding time t is recorded. Finally, the relationship between the climb height and time of the electrolyte in a specific sample is obtained. The experimental environment needs to be controlled under low humidity conditions of 2%, and the temperature can be accurately controlled by a water pot with temperature control. Results and discussion 2.1 Lucas-Washburn infiltration model Both the positive and negative electrode pieces and the diaphragm are porous structures and can be equivalent to capillary structures with different pores. The infiltration process of the electrolyte in the porous positive electrode, negative electrode and diaphragm can be understood as the capillary absorption effect. The Lucas-Washburn permeation model is used to describe the kinetics of electrode piece aspiration as shown in Equation (1).


In the formula: H is the suction height; t is the aspiration time; r- is the radius of form; σ is the surface tension of the liquid; θ is the contact angle; η is the viscosity of the liquid. It can be seen from the Lucas-Washburn equation that the square of the liquid immersion height H is proportional to the aspiration time t, and the ratio K of the square of the definition of the infiltration height H to the aspiration time t is the infiltration rate. where r- is related to the capillary shape coefficient c and the average capillary radius r, as shown in Equation (3): For the infiltration process of electrolyte in the positive and negative electrode pieces and diaphragm, the capillary shape coefficient c is positively correlated with porosity D and inversely correlated with the tortuosity τ, and the average capillary radius is the average pore size; σ and η are the surface tension and viscosity of the electrolyte, respectively; Θ is the solid-liquid two-phase contact angle, which is related to the positive/negative/diaphragm and electrolyte. 2.2 System Reliability Take 10 lithium iron phosphate cathode pieces and perform infiltration climb tests at different times in the transverse (TD) and longitudinal (MD) directions, and obtain the data shown in Figure 2. It can be found that under TD/MD in different aspiration directions, different climb heights and infiltration time t0.5 are linearly related, and the fitted R2 is 0.997, which has good accuracy. In addition, the coincidence of 5 samples in the TD direction and 5 samples in the MD direction was good, which proved that there was no difference between the TD and MD directions of the electrode piece. The overall reliability of the device is good. The relationship between immersion height and time follows the Lucas-Washburn formula. For the infiltration process of electrolyte in the positive and negative electrode pieces and diaphragm, the capillary shape coefficient c is positively correlated with porosity D and inversely correlated with the tortuosity τ, and the average capillary radius is the average pore size; σ and η are the surface tension and viscosity of the electrolyte, respectively; Θ is the solid-liquid two-phase contact angle, which is related to the positive/negative/diaphragm and electrolyte. 2.2 System Reliability Take 10 lithium iron phosphate cathode pieces and perform infiltration climb tests at different times in the transverse (TD) and longitudinal (MD) directions, and obtain the data shown in Figure 2. It can be found that under TD/MD in different aspiration directions, different climb heights and infiltration time t0.5 are linearly related, and the fitted R2 is 0.997, which has good accuracy. In addition, the coincidence of 5 samples in the TD direction and 5 samples in the MD direction was good, which proved that there was no difference between the TD and MD directions of the electrode piece. The overall reliability of the device is good. The relationship between immersion height and time follows the Lucas-Washburn formula.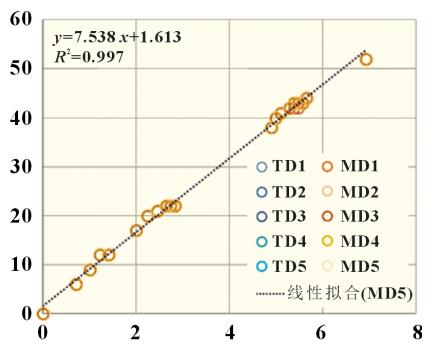 2.3 Infiltration rates of different materials The positive electrode (lithium iron phosphate), the negative electrode (artificial graphite), and the separator (PE base film) have different material and porosity characteristics. The infiltration rate of the positive/negative electrode pieces and the diaphragm were tested separately and the results are shown in Figure 3. It can be found that the negative electrode has the fastest infiltration rate, followed by the diaphragm, and the slowest is the positive electrode piece. This result shows that the porosity of the sample affects the wetting rate. The porosity of the negative electrode is usually 40%~60%, the porosity of the diaphragm and the positive electrode is 30%~40%, and the first type of pore throat has a certain relationship with the microscopic porosity of the material. 2.3 Infiltration rates of different materials The positive electrode (lithium iron phosphate), the negative electrode (artificial graphite), and the separator (PE base film) have different material and porosity characteristics. The infiltration rate of the positive/negative electrode pieces and the diaphragm were tested separately and the results are shown in Figure 3. It can be found that the negative electrode has the fastest infiltration rate, followed by the diaphragm, and the slowest is the positive electrode piece. This result shows that the porosity of the sample affects the wetting rate. The porosity of the negative electrode is usually 40%~60%, the porosity of the diaphragm and the positive electrode is 30%~40%, and the first type of pore throat has a certain relationship with the microscopic porosity of the material.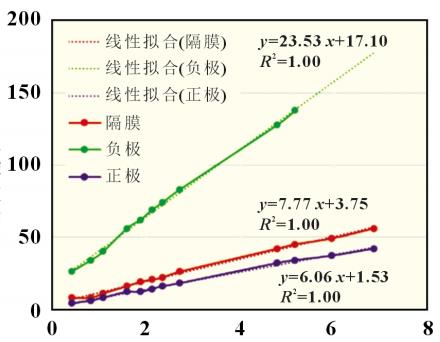 2.4 Infiltration rates for different compaction densities Piece compaction density is a key design parameter for cells. Different compaction densities correspond to different porosities and have a critical impact on battery performance. The electrolyte infiltration rate and liquid absorption volume were tested on the positive and negative electrode pieces at different compaction densities, and the results shown in Figure 4 were obtained. 2.4 Infiltration rates for different compaction densities Piece compaction density is a key design parameter for cells. Different compaction densities correspond to different porosities and have a critical impact on battery performance. The electrolyte infiltration rate and liquid absorption volume were tested on the positive and negative electrode pieces at different compaction densities, and the results shown in Figure 4 were obtained.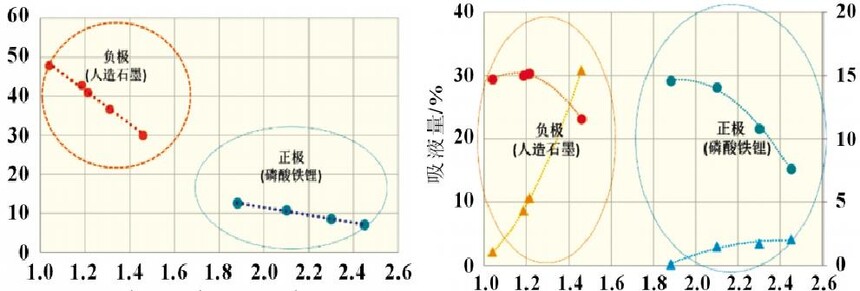 It can be found that with the increase of the compaction density of the positive and negative electrodes, the infiltration rate decreases linearly. In the range of conventional positive and negative electrode materials, the increase of the compaction density of the negative electrode is more sensitive to the influence of the wettability rate, which is about 4 times that of the positive electrode. Therefore, after the negative electrode compaction is raised, the electrolyte infiltration problem needs to be considered. In terms of saturated liquid absorption, the compaction sensitivity of lithium iron phosphate at the positive electrode is slightly greater than that of the negative electrode, about 1.5 times. In terms of swelling rate, the negative electrode with the increase of compaction density is obviously greater than the influence of the positive electrode, reaching about 10 times, which may be related to the properties of the positive and negative electrode materials and the corresponding binder. 2.5 Infiltration rates under different positive current collectors and positive formulations Under the two current collector conditions of optical foil 13μm and (13+0.5+0.5) μm double-sided coated carbon foil, the influence of different positive electrode compaction on the infiltration rate and liquid absorption volume were evaluated respectively, the positive electrode areal density was 164g/m2, and the formula ratio was the main material: binder: SP: CNT=94.5%: 3%: 1.5%: 1.5%: 1.0%. As shown in Figure 5(a), carbon foil coating is more conducive to the infiltration of electrolyte at different compaction densities. With the increase of compaction density, the gap between the wetting rate of carbon coated foil and optical foil decreases. Similar conclusions have been drawn in terms of aspiration volume, as shown in Figure 5(b). Under low pressure, the carbon coating layer can effectively increase the overall liquid absorption. With the compaction reaching a high density of 2.45g/cm3, the total liquid absorption of carbon foil and foil remains comparable. It is shown that under the condition of high pressure density, the carbon coating layer is completely embedded by the positive lithium iron phosphate, and the total liquid absorption volume is not increased. It can be found that with the increase of the compaction density of the positive and negative electrodes, the infiltration rate decreases linearly. In the range of conventional positive and negative electrode materials, the increase of the compaction density of the negative electrode is more sensitive to the influence of the wettability rate, which is about 4 times that of the positive electrode. Therefore, after the negative electrode compaction is raised, the electrolyte infiltration problem needs to be considered. In terms of saturated liquid absorption, the compaction sensitivity of lithium iron phosphate at the positive electrode is slightly greater than that of the negative electrode, about 1.5 times. In terms of swelling rate, the negative electrode with the increase of compaction density is obviously greater than the influence of the positive electrode, reaching about 10 times, which may be related to the properties of the positive and negative electrode materials and the corresponding binder. 2.5 Infiltration rates under different positive current collectors and positive formulations Under the two current collector conditions of optical foil 13μm and (13+0.5+0.5) μm double-sided coated carbon foil, the influence of different positive electrode compaction on the infiltration rate and liquid absorption volume were evaluated respectively, the positive electrode areal density was 164g/m2, and the formula ratio was the main material: binder: SP: CNT=94.5%: 3%: 1.5%: 1.5%: 1.0%. As shown in Figure 5(a), carbon foil coating is more conducive to the infiltration of electrolyte at different compaction densities. With the increase of compaction density, the gap between the wetting rate of carbon coated foil and optical foil decreases. Similar conclusions have been drawn in terms of aspiration volume, as shown in Figure 5(b). Under low pressure, the carbon coating layer can effectively increase the overall liquid absorption. With the compaction reaching a high density of 2.45g/cm3, the total liquid absorption of carbon foil and foil remains comparable. It is shown that under the condition of high pressure density, the carbon coating layer is completely embedded by the positive lithium iron phosphate, and the total liquid absorption volume is not increased.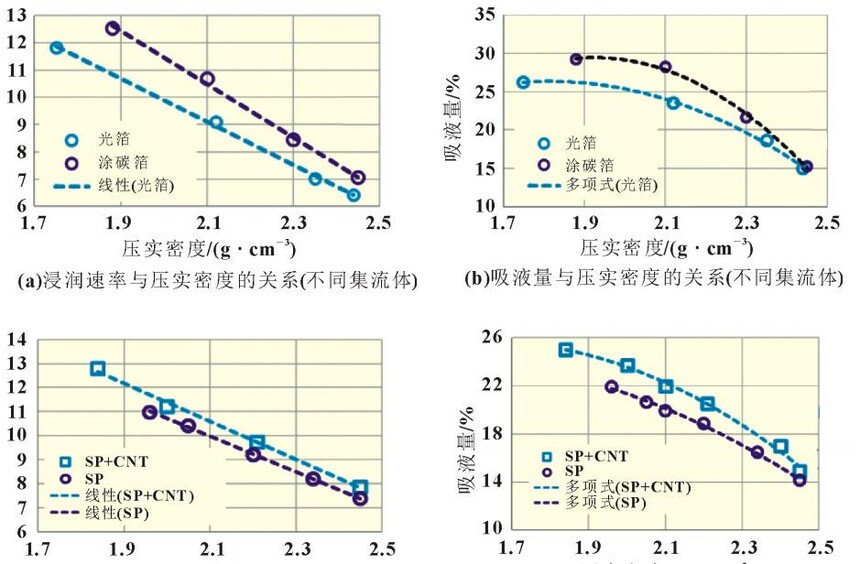 For different cathode formulations, the same binder content (3.0%) and main material content (94.5%) are used. Cathode A formulation: SP (2.5%); Positive B formula: SP (1.5%) + CNT (1.0%). The areal density of the positive electrode is 164g/m2, and the current collector is 13μm optical aluminum foil. As shown in Figure 5(c), under different compaction densities, CNT substitutes for some SP conductive agent formulations can increase the wetting rate to a certain extent. In addition, as shown in Figure 5(d), for the total aspiration volume, the SP+CNT formula has certain advantages over the SP formula under low pressure compaction, and with the increase of compaction density, the absorption volume of the CNT formula begins to increase significantly. The CNT tubular structure is also a capillary tube, and the electrolyte inside the pole piece can be remotely diffused with the help of the CNT long tube structure, thereby improving the infiltration rate of the electrolyte. However, the amount of electrolyte is mainly affected by the porosity between the particles of the main material, and with the increase of compaction, the difference in the amount of electrolyte in the remaining auxiliary materials is no longer obvious. 2.6 Infiltration rates at different temperatures A conventional 12μm PE base film was selected, and the rate of electrolyte infiltration and climbing in the diaphragm at different temperatures was tested, and the electrolyte was infiltrated in this diaphragm at different temperatures to obtain the results shown in Figure 6. It can be found that as the temperature increases, from 25 ° C to 35 and 45 ° C, the infiltration rate increases by 30% and 100%, respectively. It shows that the high temperature environment can effectively improve the wetting efficiency. This is consistent with the current idea of using high temperature immersion in industrial production.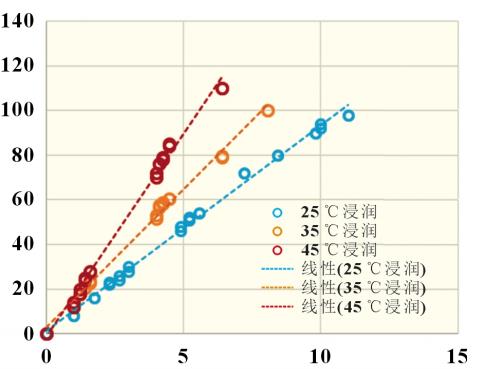 2.7 Infiltration rates under different electrolyte formulations Four electrolytes with different properties were selected, numbered as shown in Table 1. Infiltration climb within the same lithium iron phosphate cathode piece. The areal density of this positive electrode sheet is 164g/cm2 and the compaction density is 2.3g/cm3. The climb rate and electrolyte properties are shown in Figure 7, including the relationship between surface tension γ, contact angle Θ, electrolyte viscosity, η. 2.7 Infiltration rates under different electrolyte formulations Four electrolytes with different properties were selected, numbered as shown in Table 1. Infiltration climb within the same lithium iron phosphate cathode piece. The areal density of this positive electrode sheet is 164g/cm2 and the compaction density is 2.3g/cm3. The climb rate and electrolyte properties are shown in Figure 7, including the relationship between surface tension γ, contact angle Θ, electrolyte viscosity, η.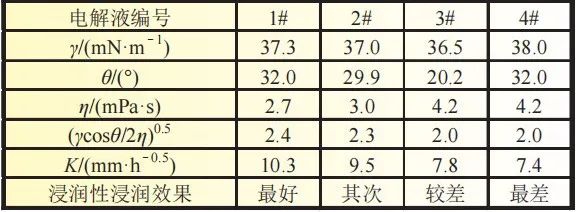 It can be found that the higher the surface tension of the electrolyte, the smaller the contact angle, the lower the viscosity, and the faster the overall infiltration rate. This conclusion fits the Lucas-Washburn formula. It is of guiding significance to match the development of highly wettable electrolytes under high-pressure dense positive electrodes. 2.8 Effect of different aspiration rates on aspiration volume and battery It can be found that the higher the surface tension of the electrolyte, the smaller the contact angle, the lower the viscosity, and the faster the overall infiltration rate. This conclusion fits the Lucas-Washburn formula. It is of guiding significance to match the development of highly wettable electrolytes under high-pressure dense positive electrodes. 2.8 Effect of different aspiration rates on aspiration volume and battery The positive and negative electrode pieces with the same areal density and different compaction density in 2.4 sections were selected, and the separator was a conventional 12μm PE separator to form a 4Ah flexible packaging laminated battery, with 26 layers of positive electrode and 27 layers of negative electrode. Firstly, the dry battery cells were subjected to aspiration experiments, and the aspiration time was 48h, and the temperature was 25 and 45 °C, respectively, to obtain the aspiration volume of different groups. Then the remaining batteries were injected, the injection amount was maintained at (15±0.5) g, and then infiltrated under different conditions (25 °C 24h, 45 °C 6h, 45 °C 12h), and finally the battery was formed and the disassembly interface was fully charged. Count the number of undesirable negative interfaces (dark spots, lithium precipitation) in different groups. Compared with different infiltration temperatures of 25 and 45 °C, the total amount of electrolyte absorption is at the same level, and the lower the positive and negative compaction density, the greater the overall liquid absorption volume. Therefore, the total liquid absorption of the battery is related to the material compaction and porosity of the pole piece itself, and external factors such as the increase of the infiltration temperature bring about the increase of the liquid absorption rate, which has no effect on the total liquid absorption volume. For the confirmation of the anode interface of the whole battery, 25 °C infiltration for 24h and 45 °C infiltration for 6 h, the negative electrodes of different groups have poor interfaces, and the number of defects is relatively low under negative very low compaction. However, the difference between the low-pressure and high-pressure groups of the positive electrode is not obvious. After the infiltration time at 45°C was increased to 12h, the interface of the other groups was good except for the negative electrode high pressure real group. It shows that the increase of the wetting rate brought by the higher wetting temperature can shorten the wetting time and improve the safety and reliability of the battery interface. Table 2 shows the statistics of the amount of liquid absorption and the number of undesirable interfaces of different groups of electrodes under different infiltration conditions. Conclusion In this paper, the key parameters affecting the aspiration rate of electrolyte were systematically studied, including the suction rate of the positive and negative electrode materials and the diaphragm itself, different positive and negative compaction densities, different current collectors and different positive electrode formulations, different infiltration temperatures and different electrolyte properties. It is found that under the conventional design, the infiltration rate of the negative electrode piece is greater than that of the positive electrode and diaphragm, and the absorption rate will decrease significantly as the compaction density of the positive and negative electrode pieces increases. The aspiration rate is more sensitive to changes in negative pressure density than changes in positive pressure density. In addition, the formulation of carbon collector and conductive carbon tube also helps to increase the positive electrode wettability rate. The increase of the infiltration temperature and the decrease of the viscosity of the electrolyte can significantly increase the aspiration rate. These are of key significance for the design and improvement of wettability of high-pressure solid electrode pieces with high energy density.
|
|




 For the infiltration process of electrolyte in the positive and negative electrode pieces and diaphragm, the capillary shape coefficient c is positively correlated with porosity D and inversely correlated with the tortuosity τ, and the average capillary radius is the average pore size; σ and η are the surface tension and viscosity of the electrolyte, respectively; Θ is the solid-liquid two-phase contact angle, which is related to the positive/negative/diaphragm and electrolyte. 2.2 System Reliability Take 10 lithium iron phosphate cathode pieces and perform infiltration climb tests at different times in the transverse (TD) and longitudinal (MD) directions, and obtain the data shown in Figure 2. It can be found that under TD/MD in different aspiration directions, different climb heights and infiltration time t0.5 are linearly related, and the fitted R2 is 0.997, which has good accuracy. In addition, the coincidence of 5 samples in the TD direction and 5 samples in the MD direction was good, which proved that there was no difference between the TD and MD directions of the electrode piece. The overall reliability of the device is good. The relationship between immersion height and time follows the Lucas-Washburn formula.
For the infiltration process of electrolyte in the positive and negative electrode pieces and diaphragm, the capillary shape coefficient c is positively correlated with porosity D and inversely correlated with the tortuosity τ, and the average capillary radius is the average pore size; σ and η are the surface tension and viscosity of the electrolyte, respectively; Θ is the solid-liquid two-phase contact angle, which is related to the positive/negative/diaphragm and electrolyte. 2.2 System Reliability Take 10 lithium iron phosphate cathode pieces and perform infiltration climb tests at different times in the transverse (TD) and longitudinal (MD) directions, and obtain the data shown in Figure 2. It can be found that under TD/MD in different aspiration directions, different climb heights and infiltration time t0.5 are linearly related, and the fitted R2 is 0.997, which has good accuracy. In addition, the coincidence of 5 samples in the TD direction and 5 samples in the MD direction was good, which proved that there was no difference between the TD and MD directions of the electrode piece. The overall reliability of the device is good. The relationship between immersion height and time follows the Lucas-Washburn formula. 2.3 Infiltration rates of different materials The positive electrode (lithium iron phosphate), the negative electrode (artificial graphite), and the separator (PE base film) have different material and porosity characteristics. The infiltration rate of the positive/negative electrode pieces and the diaphragm were tested separately and the results are shown in Figure 3. It can be found that the negative electrode has the fastest infiltration rate, followed by the diaphragm, and the slowest is the positive electrode piece. This result shows that the porosity of the sample affects the wetting rate. The porosity of the negative electrode is usually 40%~60%, the porosity of the diaphragm and the positive electrode is 30%~40%, and the first type of pore throat has a certain relationship with the microscopic porosity of the material.
2.3 Infiltration rates of different materials The positive electrode (lithium iron phosphate), the negative electrode (artificial graphite), and the separator (PE base film) have different material and porosity characteristics. The infiltration rate of the positive/negative electrode pieces and the diaphragm were tested separately and the results are shown in Figure 3. It can be found that the negative electrode has the fastest infiltration rate, followed by the diaphragm, and the slowest is the positive electrode piece. This result shows that the porosity of the sample affects the wetting rate. The porosity of the negative electrode is usually 40%~60%, the porosity of the diaphragm and the positive electrode is 30%~40%, and the first type of pore throat has a certain relationship with the microscopic porosity of the material. 2.4 Infiltration rates for different compaction densities Piece compaction density is a key design parameter for cells. Different compaction densities correspond to different porosities and have a critical impact on battery performance. The electrolyte infiltration rate and liquid absorption volume were tested on the positive and negative electrode pieces at different compaction densities, and the results shown in Figure 4 were obtained.
2.4 Infiltration rates for different compaction densities Piece compaction density is a key design parameter for cells. Different compaction densities correspond to different porosities and have a critical impact on battery performance. The electrolyte infiltration rate and liquid absorption volume were tested on the positive and negative electrode pieces at different compaction densities, and the results shown in Figure 4 were obtained. It can be found that with the increase of the compaction density of the positive and negative electrodes, the infiltration rate decreases linearly. In the range of conventional positive and negative electrode materials, the increase of the compaction density of the negative electrode is more sensitive to the influence of the wettability rate, which is about 4 times that of the positive electrode. Therefore, after the negative electrode compaction is raised, the electrolyte infiltration problem needs to be considered. In terms of saturated liquid absorption, the compaction sensitivity of lithium iron phosphate at the positive electrode is slightly greater than that of the negative electrode, about 1.5 times. In terms of swelling rate, the negative electrode with the increase of compaction density is obviously greater than the influence of the positive electrode, reaching about 10 times, which may be related to the properties of the positive and negative electrode materials and the corresponding binder. 2.5 Infiltration rates under different positive current collectors and positive formulations Under the two current collector conditions of optical foil 13μm and (13+0.5+0.5) μm double-sided coated carbon foil, the influence of different positive electrode compaction on the infiltration rate and liquid absorption volume were evaluated respectively, the positive electrode areal density was 164g/m2, and the formula ratio was the main material: binder: SP: CNT=94.5%: 3%: 1.5%: 1.5%: 1.0%. As shown in Figure 5(a), carbon foil coating is more conducive to the infiltration of electrolyte at different compaction densities. With the increase of compaction density, the gap between the wetting rate of carbon coated foil and optical foil decreases. Similar conclusions have been drawn in terms of aspiration volume, as shown in Figure 5(b). Under low pressure, the carbon coating layer can effectively increase the overall liquid absorption. With the compaction reaching a high density of 2.45g/cm3, the total liquid absorption of carbon foil and foil remains comparable. It is shown that under the condition of high pressure density, the carbon coating layer is completely embedded by the positive lithium iron phosphate, and the total liquid absorption volume is not increased.
It can be found that with the increase of the compaction density of the positive and negative electrodes, the infiltration rate decreases linearly. In the range of conventional positive and negative electrode materials, the increase of the compaction density of the negative electrode is more sensitive to the influence of the wettability rate, which is about 4 times that of the positive electrode. Therefore, after the negative electrode compaction is raised, the electrolyte infiltration problem needs to be considered. In terms of saturated liquid absorption, the compaction sensitivity of lithium iron phosphate at the positive electrode is slightly greater than that of the negative electrode, about 1.5 times. In terms of swelling rate, the negative electrode with the increase of compaction density is obviously greater than the influence of the positive electrode, reaching about 10 times, which may be related to the properties of the positive and negative electrode materials and the corresponding binder. 2.5 Infiltration rates under different positive current collectors and positive formulations Under the two current collector conditions of optical foil 13μm and (13+0.5+0.5) μm double-sided coated carbon foil, the influence of different positive electrode compaction on the infiltration rate and liquid absorption volume were evaluated respectively, the positive electrode areal density was 164g/m2, and the formula ratio was the main material: binder: SP: CNT=94.5%: 3%: 1.5%: 1.5%: 1.0%. As shown in Figure 5(a), carbon foil coating is more conducive to the infiltration of electrolyte at different compaction densities. With the increase of compaction density, the gap between the wetting rate of carbon coated foil and optical foil decreases. Similar conclusions have been drawn in terms of aspiration volume, as shown in Figure 5(b). Under low pressure, the carbon coating layer can effectively increase the overall liquid absorption. With the compaction reaching a high density of 2.45g/cm3, the total liquid absorption of carbon foil and foil remains comparable. It is shown that under the condition of high pressure density, the carbon coating layer is completely embedded by the positive lithium iron phosphate, and the total liquid absorption volume is not increased.
 2.7 Infiltration rates under different electrolyte formulations Four electrolytes with different properties were selected, numbered as shown in Table 1. Infiltration climb within the same lithium iron phosphate cathode piece. The areal density of this positive electrode sheet is 164g/cm2 and the compaction density is 2.3g/cm3. The climb rate and electrolyte properties are shown in Figure 7, including the relationship between surface tension γ, contact angle Θ, electrolyte viscosity, η.
2.7 Infiltration rates under different electrolyte formulations Four electrolytes with different properties were selected, numbered as shown in Table 1. Infiltration climb within the same lithium iron phosphate cathode piece. The areal density of this positive electrode sheet is 164g/cm2 and the compaction density is 2.3g/cm3. The climb rate and electrolyte properties are shown in Figure 7, including the relationship between surface tension γ, contact angle Θ, electrolyte viscosity, η. It can be found that the higher the surface tension of the electrolyte, the smaller the contact angle, the lower the viscosity, and the faster the overall infiltration rate. This conclusion fits the Lucas-Washburn formula. It is of guiding significance to match the development of highly wettable electrolytes under high-pressure dense positive electrodes. 2.8 Effect of different aspiration rates on aspiration volume and battery
It can be found that the higher the surface tension of the electrolyte, the smaller the contact angle, the lower the viscosity, and the faster the overall infiltration rate. This conclusion fits the Lucas-Washburn formula. It is of guiding significance to match the development of highly wettable electrolytes under high-pressure dense positive electrodes. 2.8 Effect of different aspiration rates on aspiration volume and battery